Nepheir
Finding out you need a need a kidney transplant is daunting but the search for one shouldn't be.

I met Varun through Reddit, and we began working on this platform together, inspired by the journey of his father.

Background
In the U.S., 1 in 7 adults live with chronic kidney disease (CKD). In 2023 alone, 46,000 kidney transplants were performed. Sadly, more than 104,000 people are on the national waitlist, and many do not receive a transplant in time due to limited access to information regarding transplant programs.



Timeline: April 2024 - July 2024
Tools: Figma / Maze / Discord
Role: UX & UI Designer

Goals
1
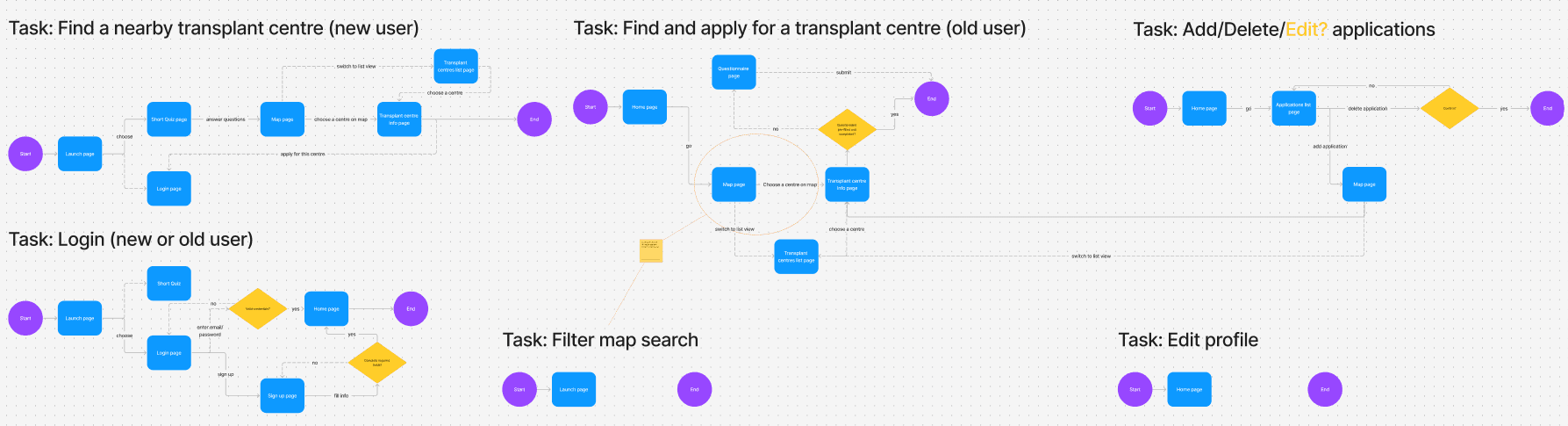
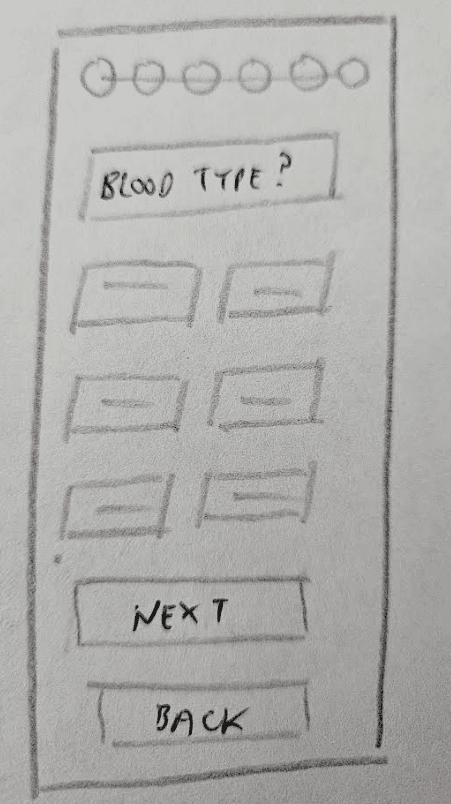
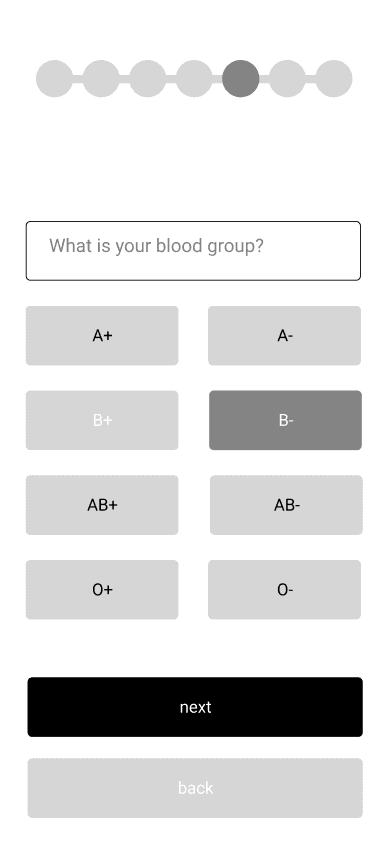
Incorporate essential, commonly asked patient questions into the user experience that are neither intrusive nor vague.
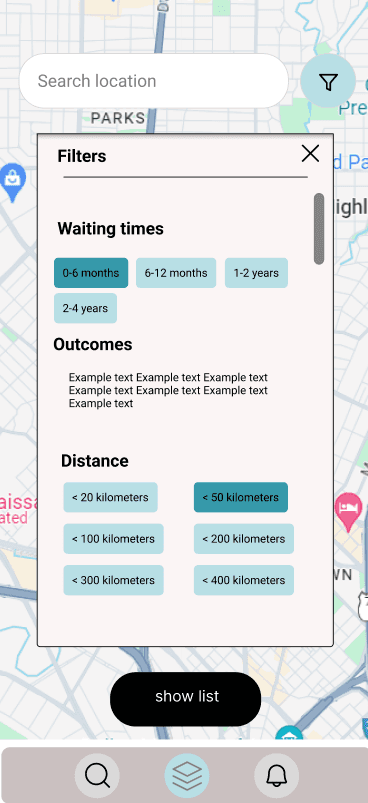
The listing process requires prospective transplant recipients to answer the same series of questions. Thoughtfully integrating this step into the user experience will be one of three key focus areas.
2
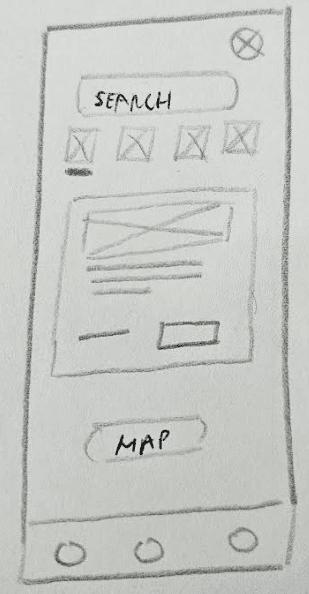
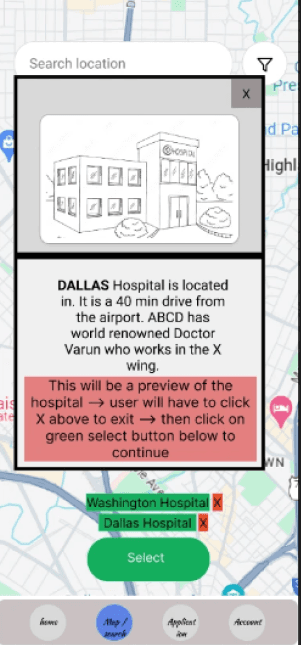
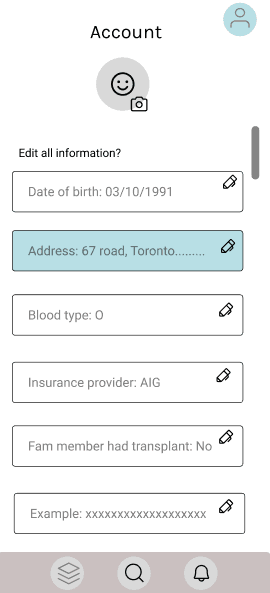
Effectively present extensive and complex information in a user-friendly manner.
The integration of these advanced technologies is reshaping how we approach design, offering new possibilities and challenges.
3
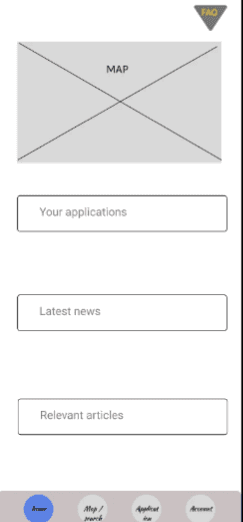
Create a seamless user interface that enhances the overall process.
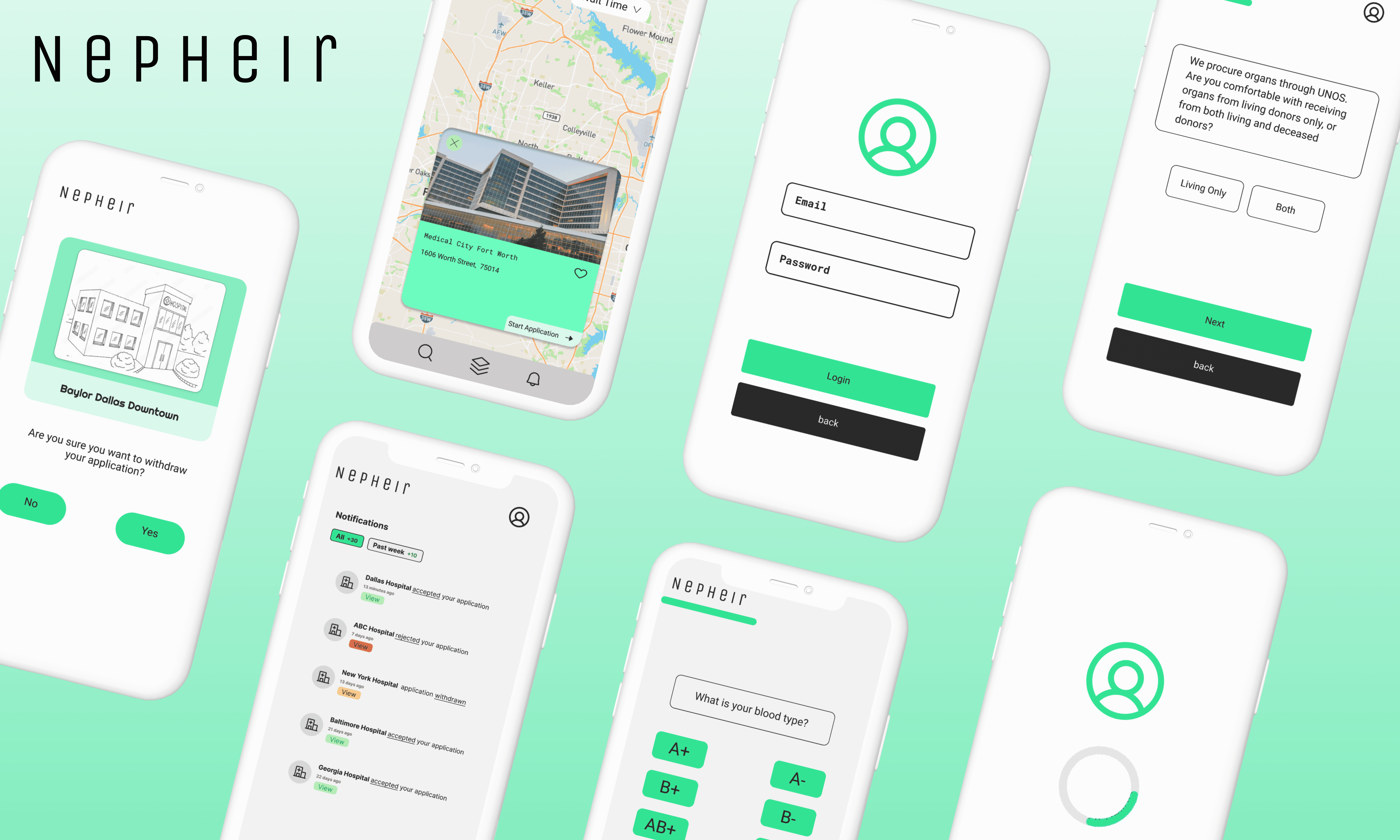
I wanted to create a UI that both enhances and simplifies the process of Multi-Listing.

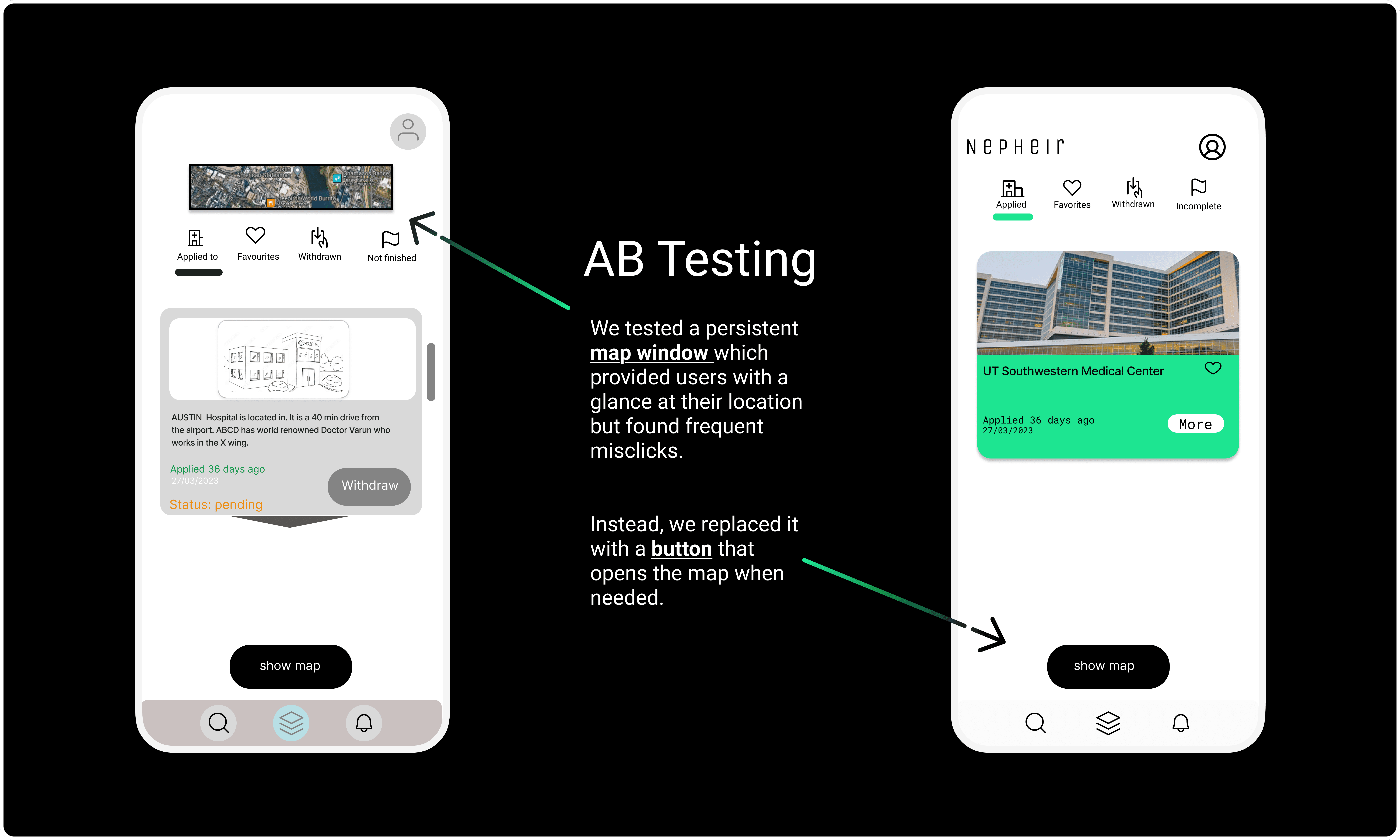
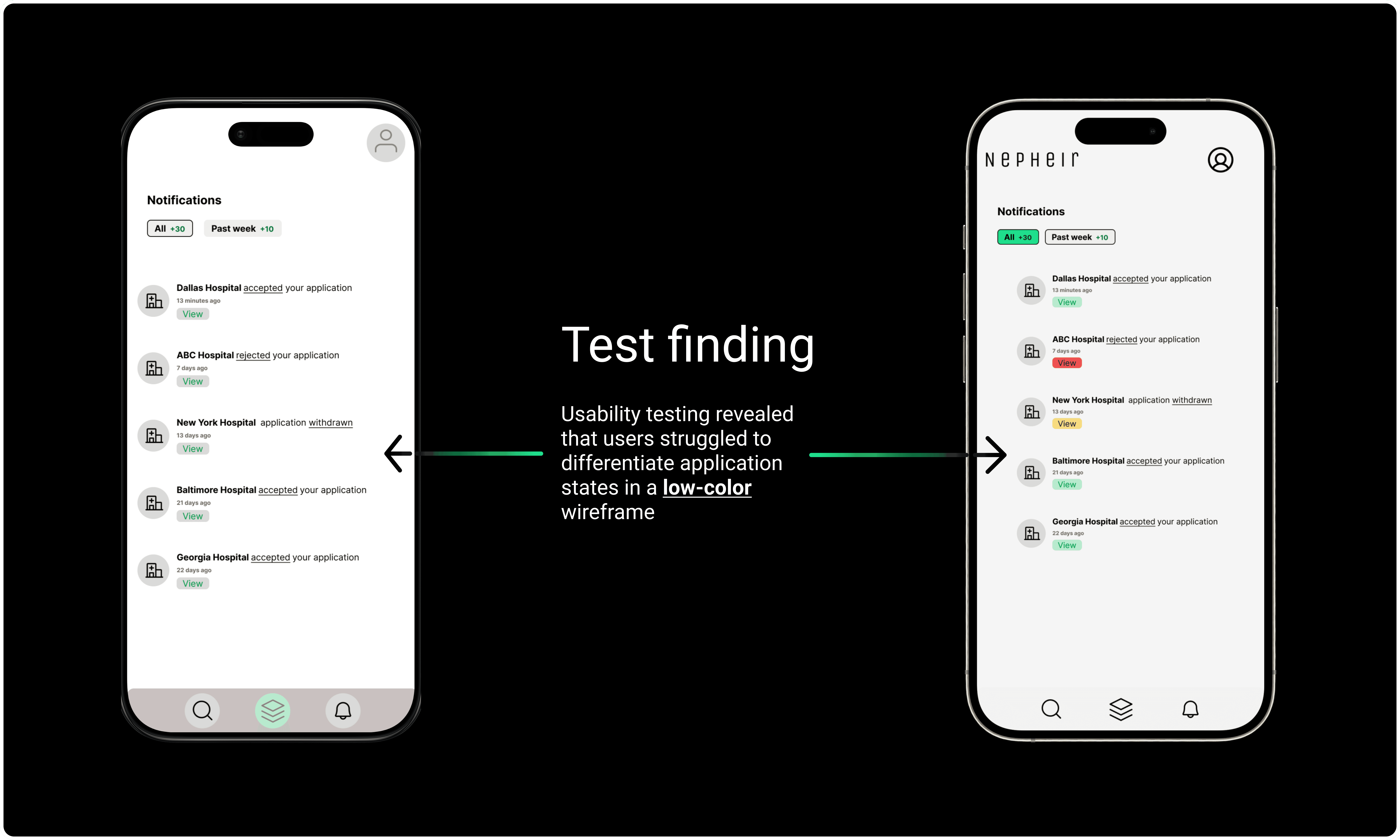
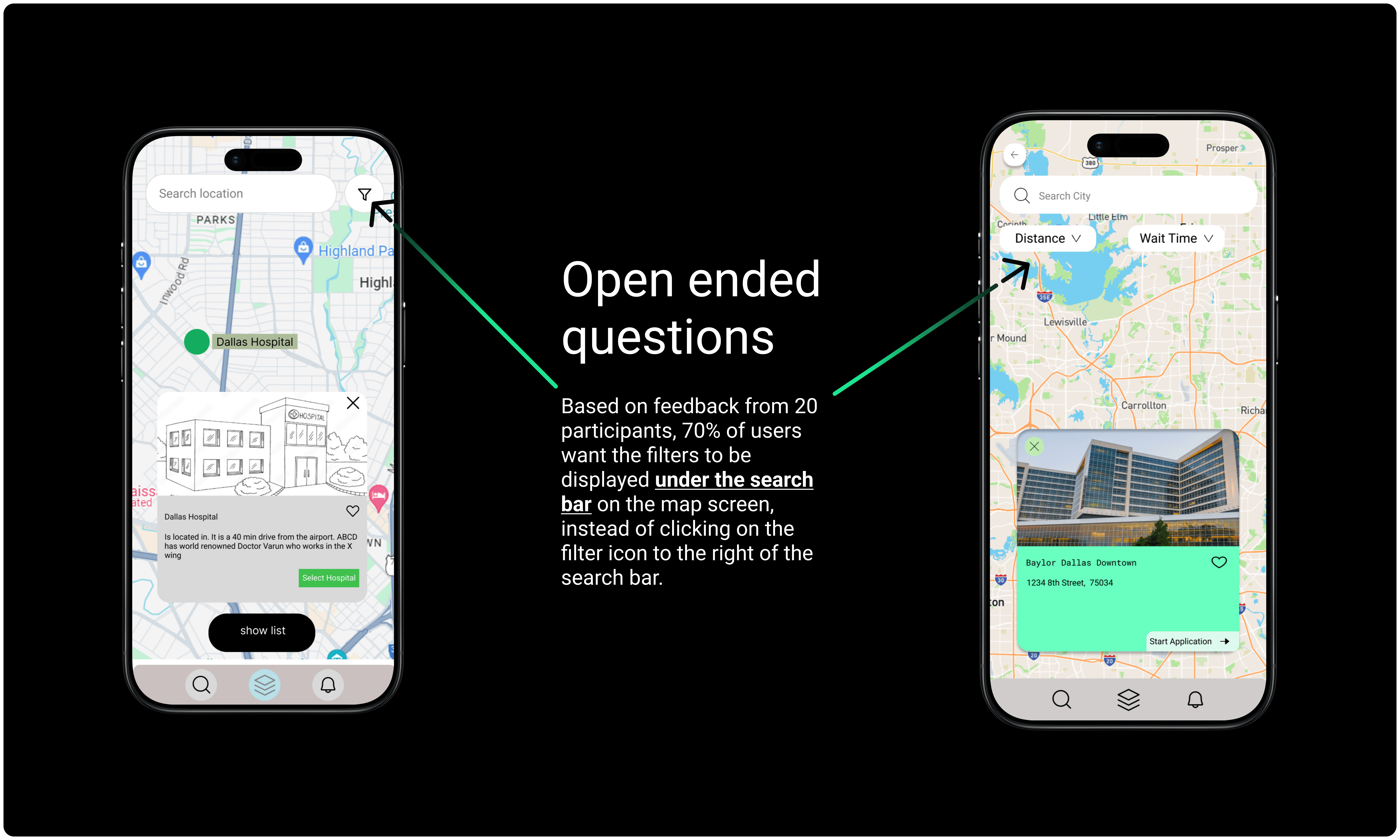
Conclusion
Our design decisions for Nepheir were shaped by a team member’s personal experiences and the challenges faced by transplant recipients. We prioritized user-centric design with seamless interaction and clear navigation to simplify complex medical information.
This was my first end-to-end UX project, giving me a strong grasp of MVP development. Although healthcare isn’t my primary field, the experience boosted my confidence and prepared me to take on future case studies independently.
Working with Varun was a highlight—we stayed on schedule and completed the project in ten weeks.
If I could start over, I’d build the MVP earlier to allow for user testing by week three, giving us more time to refine the design.